Cardiac Safety Studies
Ion Channel Assays
- hERG screening (early identification of pro-arrhythmic effects)
- hERG, Nav1.5 and Cav1.2, Cav3.2, Kir2.1
- Large cardiac panel: hERG, Nav1.5, Cav1.2, Cav3.2, Kir2.1, Kv4.3/KChIP and Kv7.1/MinK
hiPSC Cardiomyocyte Assays
-
Action potential prolongation and early afterdepolarizations by hERG inhibitors.
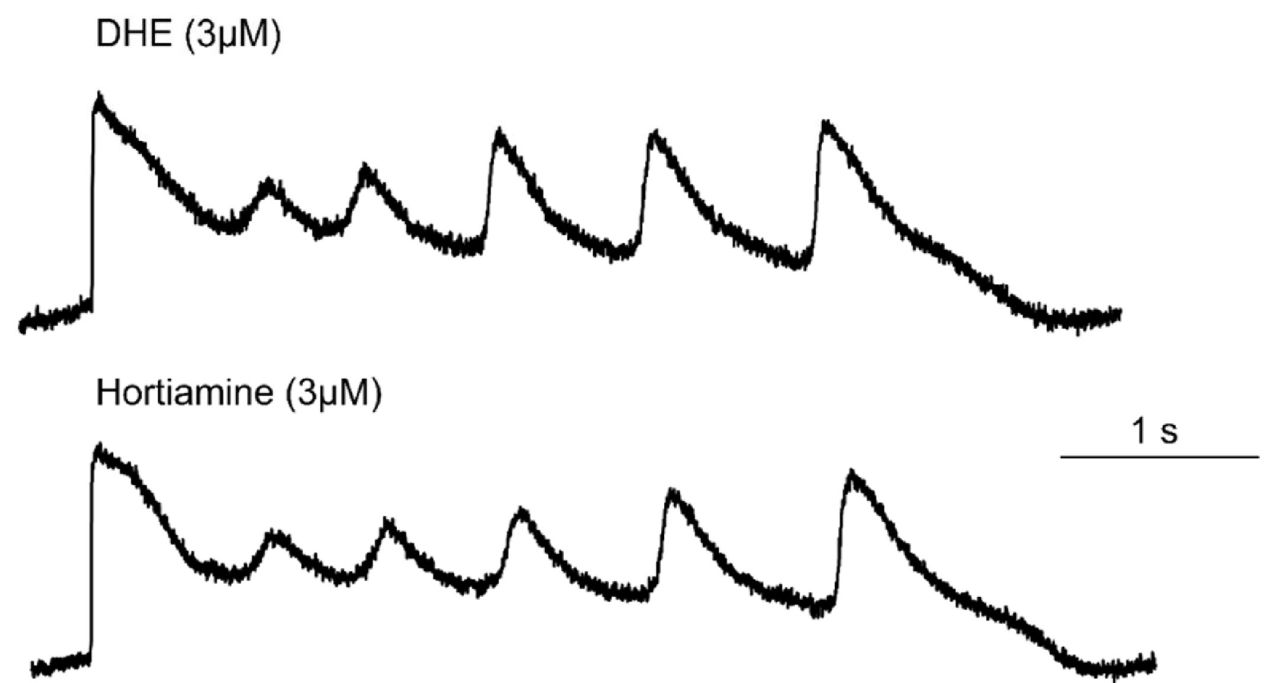
Figure: Effects of the natural products hortiamine and dehydroevodiamine (DHE) on action potentials (FluoVolt™) on hiPSC-CM (Baburin et al. 2018). - Field potential parameters (R(Q)-T interval, others) of hiPSC in 2D cultures assessed with Multi-Electrode Arrays
- Field potential parameters (R(Q)-T interval, others) of hiPSC in 3D cardiospheres assessed with Multi-Electrode Arrays
- Current clamp recordings on hiPSC to assess action potential parameters — Manual Patch Clamp (gold standard, highest resolution)
Technologies
Our services are supported by industry-recognized platforms commonly used in pharmaceutical research for comprehensive safety profiling.
SyncroPatch 384
High-throughput cardiac safety screening.
The SyncroPatch 384 allows for the rapid assessment of compound effects on cardiac ion channels (e.g., hERG, Nav1.5, Cav1.2) in compliance with CiPA guidelines. It enables the generation of large datasets to identify cardiac liabilities early in the drug discovery process.
Best suited for: Early hazard identification, hERG potency ranking, hit-to-lead safety profiling
NANION TECHNOLOGIES HIGH THROUGHPUT
Patchliner
Versatile automated safety profiling.
With precise temperature control and high seal resistance, the Patchliner is ideal for in-depth characterization of drug-channel interactions. It supports complex voltage protocols required for detailed kinetic analysis and mechanism-of-action studies.
Best suited for: In-depth safety profiling, temperature-dependent assays, detailed mechanism analysis
NANION TECHNOLOGIES MEDIUM THROUGHPUT
Manual Patch Clamp
The reference standard for cardiac electrophysiology.
Manual patch clamp offers the highest fidelity for recording Action Potentials (APs) from iPSC-derived cardiomyocytes. This approach allows for the detection of Early Afterdepolarizations (EADs) and other pro-arrhythmic markers that automated systems might miss.
Best suited for: Action potential profiling, validation of automated data, late-stage safety assessment
GOLD STANDARD CIPA COMPLIANT
MEA Systems (Multi Channel Systems)
Network-level functional assessment.
Microelectrode Array (MEA) technology records extracellular field potentials from cardiomyocyte monolayers, capturing arrhythmia-like events and conduction velocities in a physiological network context.
Best suited for: Pro-arrhythmia prediction, beat rate variability, chronic drug exposure studies
MCS PHYSIOLOGICAL RELEVANCE
High-Speed sCMOS Imaging (Kinetix)
High-speed optical phenotyping.
Using the Kinetix sCMOS camera, we capture rapid calcium transients and voltage changes in large populations of cardiomyocytes with exceptional temporal resolution, allowing for the detection of subtle wavefront propagation abnormalities.
Best suited for: Calcium handling dynamics, wavefront propagation analysis
PHOTOMETRICS HIGH SPEED
Contractility & Impedance Analysis
Excitation-contraction coupling assessment.
We assess the physical beating properties of cardiomyocytes (amplitude, relaxation time) to detect compounds that affect cardiac contractility (inotropes/lusitropes) or cause structural toxicity.
Best suited for: Cardiotoxicity assessment, structural integrity monitoring, contractility modulation
In-silico Pro-arrhythmia Modeling
Translational risk prediction.
Our computational modeling integrates in vitro ion channel data to predict clinical pro-arrhythmic risk. This bridges the gap between assay results and human safety profiles, providing a quantitative risk score.
Best suited for: CiPA risk scoring, early candidate selection, data integration
COMPUTATIONAL BIOLOGY PREDICTIVE