Advancing Cardiac Safety Studies
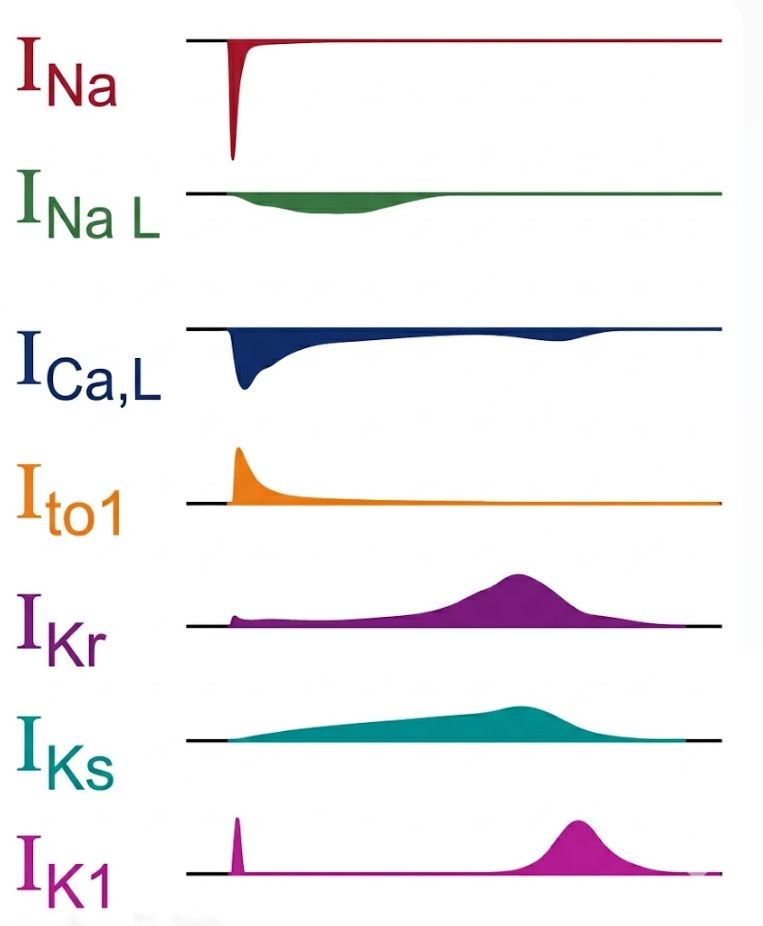
ChanPharm provides in vitro proarrhythmia assays on cardiac ion channels with different technologies:
- High-throughput screening (HTS) on small and large panel of cardiac ion channels
- Manual patch clamp on selected cardiac ion channels
- Studies on Human induced pluripotent stem cell-derived cardiomyocytes (hiPSC-CMs) with Multi Electrode arrays (MEAs)
- Studies on hiPSC with voltage sensitive dyes (VSD)
If necessary, comparative studies are conducted using MEA technology and VSD, which further increases the quality and significance of these studies.
Potential cardiotoxic effects can be conclusively analyzed using various in silico models.
Cardiac Channel Panel
| CIPA Ion Channels | Target | Platform | |
|---|---|---|---|
| Manual Patch | Syncropatch | ||
| Small Cardiac Panel | hERG | ||
| hNaV1.5 | |||
| hCaV1.2 | |||
| hCaV3.2 | |||
| hKV1.5 | |||
| hKV7.1_minK | |||
| +Extended Cardiac Panel | hKir2.1 | ||
| hKir3.1 | |||
| hKir3.4 | |||
| hKV4.3/KChip2.2 | |||
| HCN2 | |||
| HCN4 | |||
Making use of our assays against hERG, NaV1.5, CaV1.2, CaV3.2, Kv7.1/minK and Kv1.5 (small cardiac panel) we detect potential proarrhythmic drug effects during early stages of drug development. These assays are particularly important for assessing drug-induced arrhythmic risks.
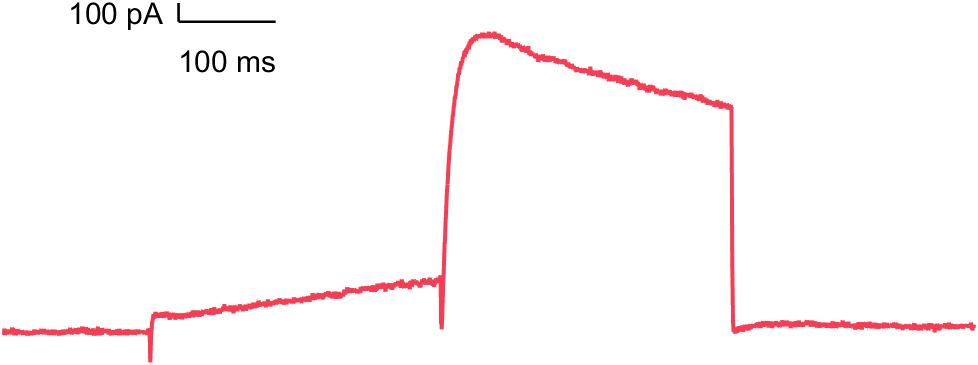
hERG (Kv11.1)
Inhibition of this ion channel may induce a delay in the repolarization of the cardiac action potential and cause severe arrhythmia (FDA S7B guidance).
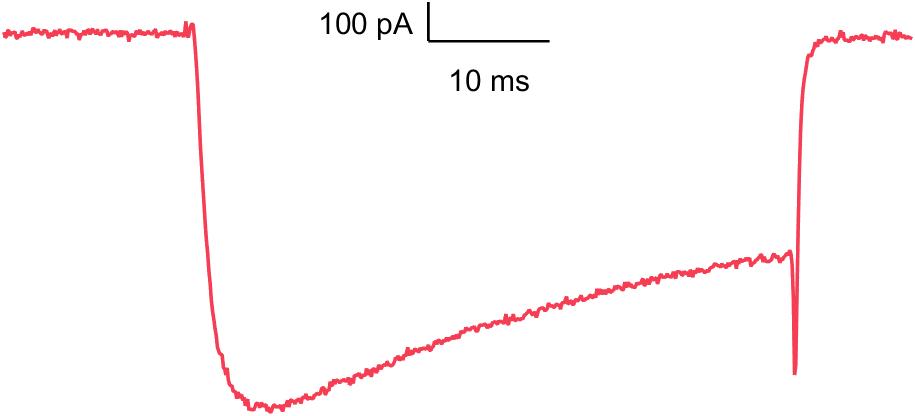
CaV1.2
Blockade of this calcium channel can shorten the QT interval and causes negative inotropic effects.
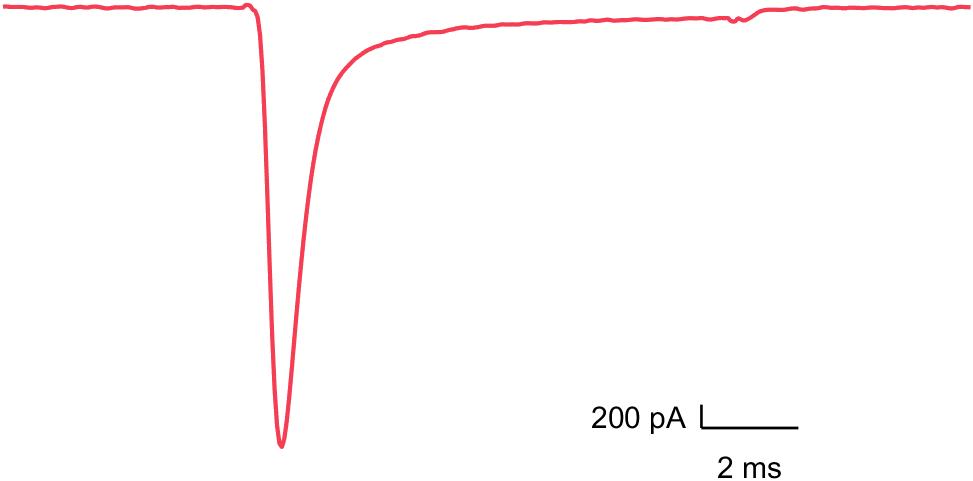
NaV1.5
Block of this cardiac sodium channel reduces conduction velocity leading to different forms of arrhythmia.
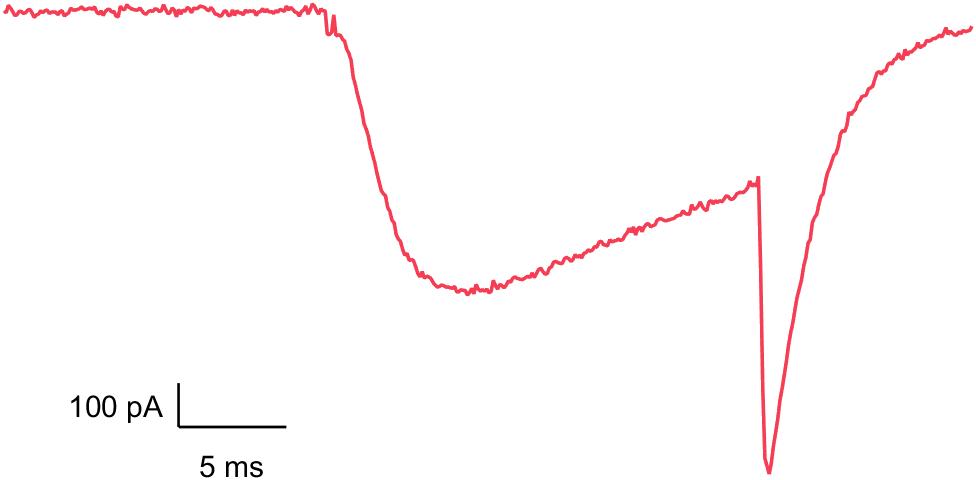
CaV3.2
Inhibition of these T-type calcium channels can cause bradycardia or neurological impairments.

Kv7.1/minK
Inhibition can cause severe arrhythmia such as Long QT Syndrome (LQTS) and Torsades de Pointes (TdP).
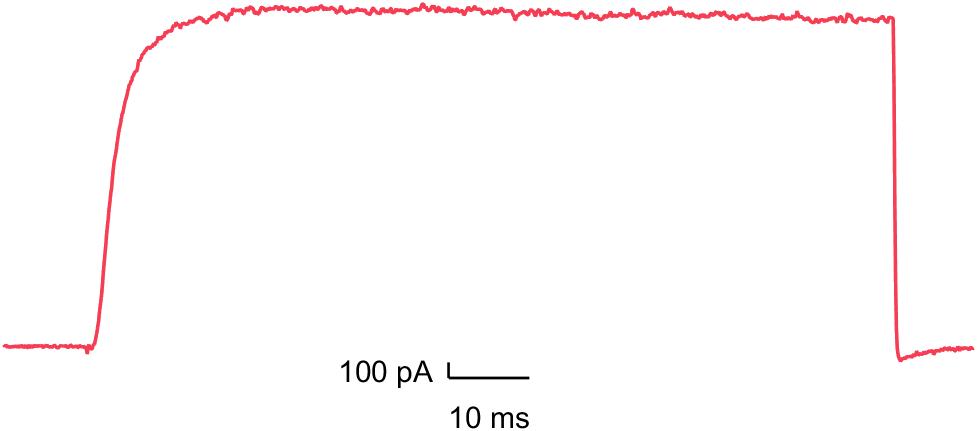
Kv1.5
Inhibition can cause different forms of arrhythmia such as QT prolongation and neuronal hyperexcitability.