Validation Data & Case Studies
We have conducted extensive pharmacological profiling to validate the physiological relevance of our preclinical models. Below are representative manual patch clamp recordings from our screening platforms.
1. Current clamp studies on human iPSC-derived sensory neurons
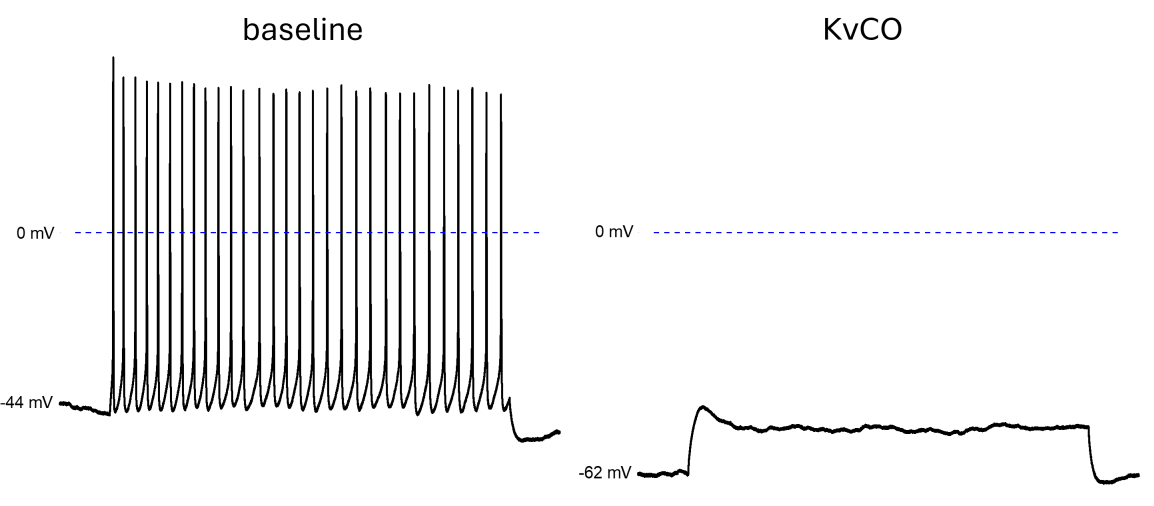
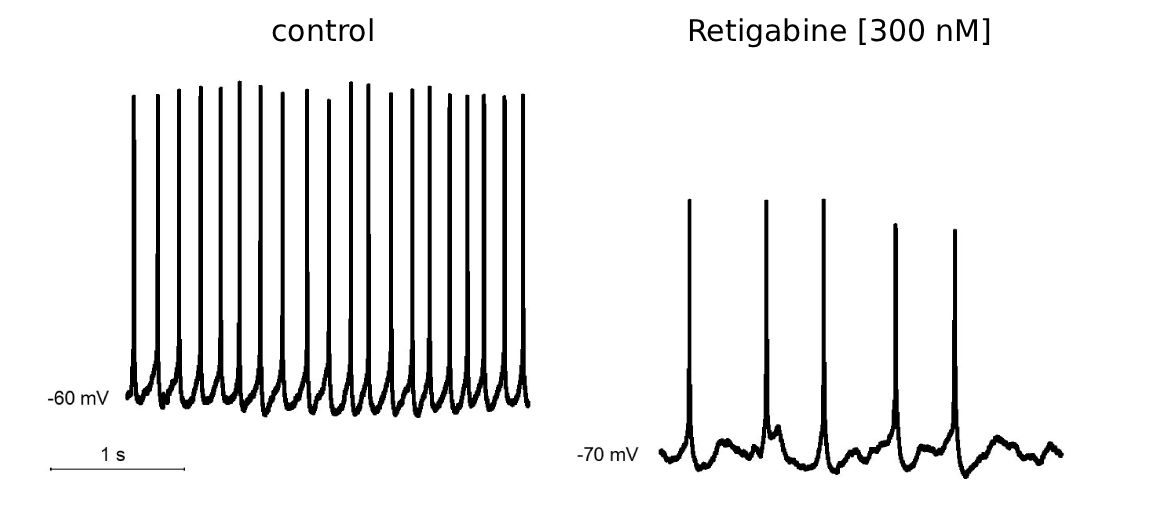
Current clamp of human iPSC-derived sensory neurons exhibit robust spontaneous repetitive firing. Application of 10 µM of a Kv7.2/3 channel opener (KvCO) suppressed repetitive action potential firing. Similarly, 300 nM Retigabine significantly reduces firing frequency and hyperpolarizes the resting membrane potential.
2. Voltage clamp studies on human iPSC-derived sensory neurons
Key Findings: Characterization of Tetrodotoxin (TTX)-sensitive and Nav1.7-mediated sodium currents using selective blockers.
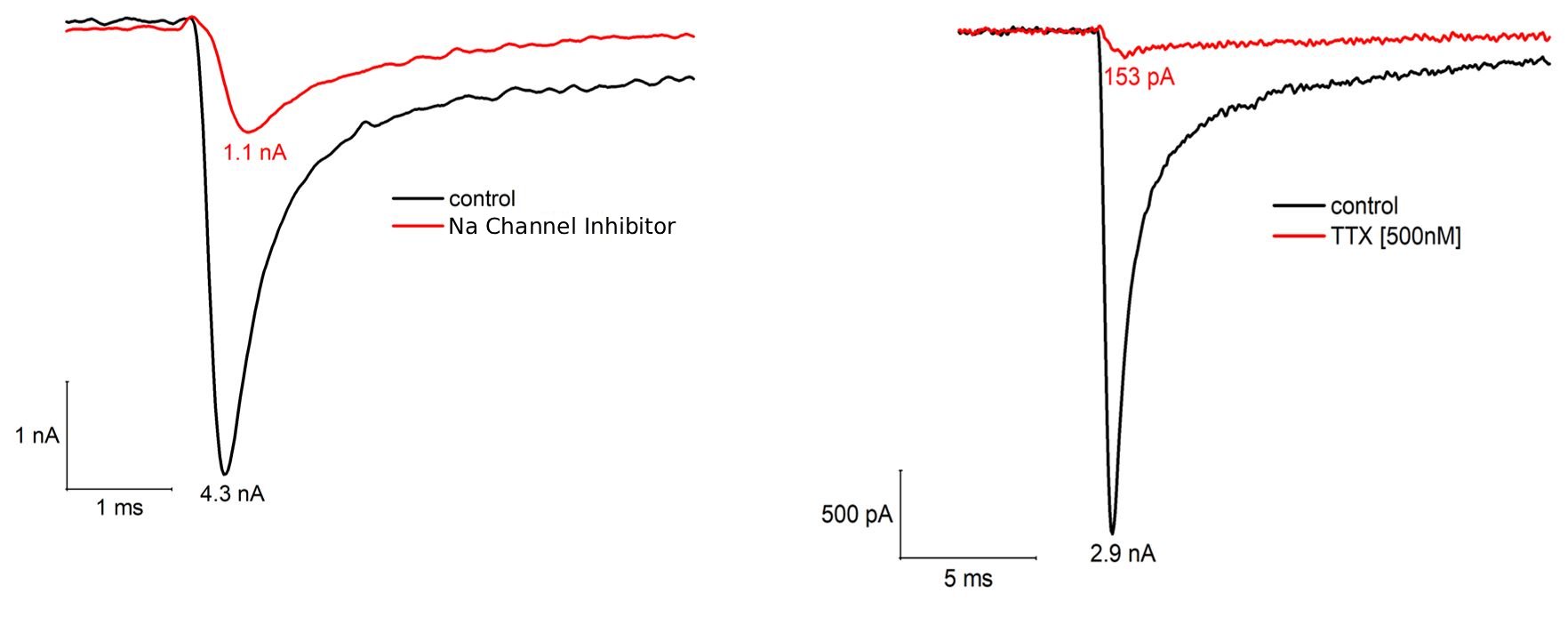
Pharmacology: The fast inward current is suppressed by TTX (500 nM) and a selective Nav1.7 inhibitor. This pharmacological profile confirms a predominance of TTX-sensitive isoforms, specifically identifying Nav1.7 as a primary functional component in this sensory neuron model.
3. Primary Dorsal Root Ganglion (DRG) neurons (rat)
Key Findings: Identification of Nav1.8-mediated TTX-resistant (TTX-R) sodium currents and validation with clinical standards.
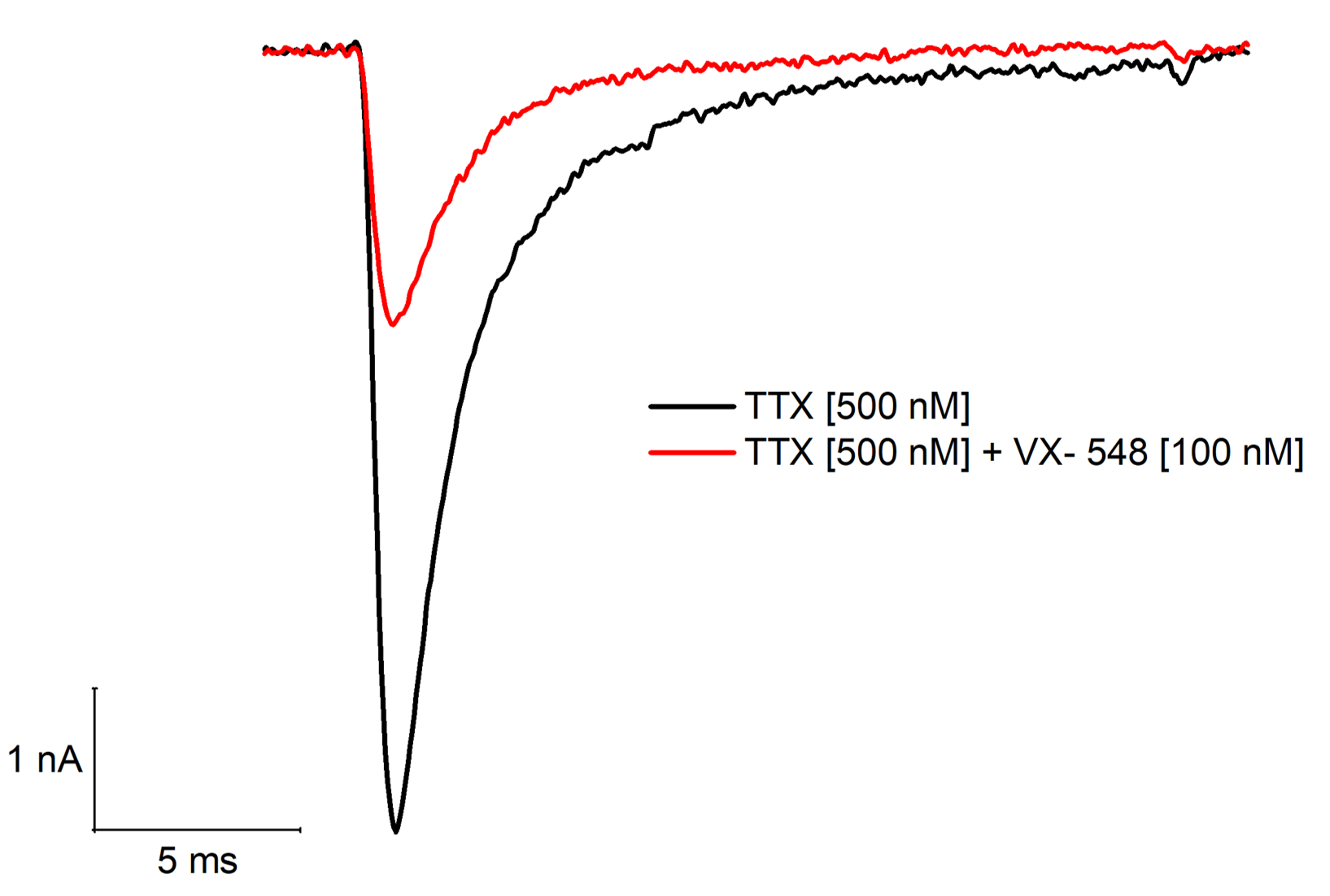
A TTX-resistant fast inward current (black trace) is substantially inhibited by 100 nM of the highly selective Nav1.8 inhibitor VX-548 (Suzetrigine).
Clinical Validation: Application of 100 nM VX-548 (Suzetrigine)—a highly selective, clinically validated Nav1.8 inhibitor—resulted in a potent block of the remaining current (red trace). This demonstrates our platform's capability to accurately screen and validate compounds against native pain targets in their physiological environment.